Sign Up
ONWARD requests this information to keep you updated on the progress we are making to develop meaningful innovations that are designed to improve the lives of people with spinal cord injury.
Please review our Privacy Policy for more information on how we use and disclose your information.
The personal data collected is intended for ONWARD and used to process your request and to send you information emails if you have consented to it. For more information click here
Mandatory Data are indicated on the form by an asterisk. Access to the Data is strictly limited to the employees of ONWARD in charge of processing your request.
In accordance with Regulation (EU) 2016/679 of 27 April 2016 on the protection of personal Data, you have the right of access, rectification, erasure, portability and limitation of the Processing of your Personal Data.
You also have the right to communicate directives on the fate of your Data after your death.
You also have the right to object to the Processing of your Data. You may exercise your rights by contacting the DPO at the following address privacy@onwd.com or at the following postal address: 3 Pont Bessières 1005 Lausanne, Switzerland.
For more information on the Processing of your Personal Data by ONWARD, please consult our privacy policy available on our website at the following address: https://www.onwd.com/privacy-policy

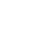





























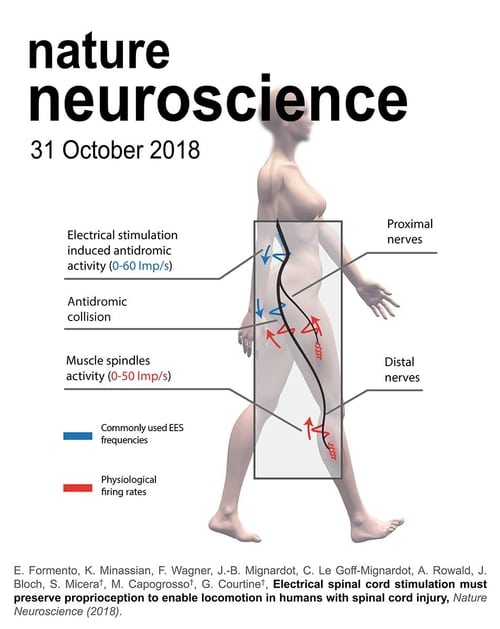
























%20(1).png)














.png)



.png)




































